Blog provides NCERT solutions, CBSE, NTSE, Olympiad study material, model test papers, important Questions and Answers asked in CBSE examinations. References to Educational Sites and resources.
Showing posts with label class8-science. Show all posts
Showing posts with label class8-science. Show all posts
Saturday 5 August 2017
Friday 21 July 2017
CBSE Class 8 - General Science - Unit Test Paper - 1 (2017-18) (#cbsePapers)
CBSE Class 8 - General Science - Unit Test Paper - 1 (2017-18)
Chapters covered:1. Microorganisms
2. Metals and Non-Metals
3. Force and Pressure

Wednesday 12 July 2017
CBSE Class 9 - Force and Laws of Motion - What is Inertia? (#cbseNotes)
What is Inertia?
CBSE Class 9 - Force and Laws of Motion
 |
| Let's move the rock! |
Inertia
It is inherent property of all bodies, by virtue of which it resists or opposes any change to it's present state of rest or of uniform motion. It varies directly as mass of the body.
Inertia of a body is of three types:
① Inertia of rest
It is in ability of a body to change by itself or if state of rest. This means a body at rest remains at rest and cannot start moving on its own.
Tuesday 11 July 2017
CBSE Class 8 - Science Periodic Test-1 (2017-18) (#cbseNotes)
CBSE Class 8 - Science Periodic Test-1 (2017-18)
Questions asked in the Q. Paper are from the following chapters:1. Crop Production and Management
2. Coal and Petroleum
3. Combustion and Flame
4. Universe

CBSE Class 8 - Science - Synthetic Fibres - Know Your Plastic Bottle (#cbseNotes)
Know Your Plastic Bottle
Class 8 - Science - Synthetic Fibres
 |
| Resin Coding System (Image Credits: Openclipart) |
In 1988, the Plastic Industry Association developed the Resin Identification Codes (1-7), a set of symbols appearing on plastic products that identify the plastic resin out of which the product is made. This help the workers to sort out various plastic bottles in order to recyle or reuse them or not.
Here is a table of resin codes that are used to get information about the application of plastic, its safety and re-usability.
♳ PET/PETE
Polymer: Polythene Terephtalate
Common Applications: Soft Drink, Water and Salad Dressing bottles, peanut butter and jam jars.
Safe for Alimentary Use: Safe One Type Use.
Reusable: No

Wednesday 5 July 2017
CBSE Class 8 - Science Periodic Assessment (2017) - Crop Production and Microorganisms (#cbseNotes)
CBSE Class 8 - Science Periodic Assessment (2017) - Crop Production and Microorganisms
Sample Question Paper with some answers given below.

Monday 3 July 2017
CBSE Class 8 - Science - Friction (Assignment) (#cbseNotes)
Friction (Assignment)
Q1: Name the following:
⒜ Type of surfaces that produces minimum friction _____________
⒝ Type of surfaces that produce maximum friction _____________
⒞ A shape that reduces drag _____________
⒟ Friction with minimum magnetic magnitude _____________
⒠ Friction with maximum magnitude _____________
⒡ Two types of fluids _____________

Thursday 29 June 2017
CBSE Class 8 - Science - Friction (MCQs) (#cbseNotes)
Friction (MCQs)
CBSE Class 8 - Science
Q1: The force of friction which opposes the surfaces just to slide one over the other, is called:
(A) Limiting friction.
(B) Rolling friction
(C) Sliding friction
(D) All of these.
Q2: What is the most common way to reduce friction?
(A) Lubrication.
(B) Warning signs.
(C) Eliminate the motion.
(D) None of these.
Q3: Do you want to increase or decrease friction when riding a bicycle?
(A) Increase.
(B) Both.
(C) Decrease.
(D) None of these.

Monday 26 June 2017
CBSE Class 8 – Science – Microorganisms – Friends or Foes (Worksheet) (#cbseNotes)
Microorganisms – Friends or Foes (Worksheet)
1. Study of Microorganisms : __________________
2. The size of bacterium is 3.5 microns = ___________ mm = ___________ m
3. Microbes are divided into four categories:
i. _____________ ii. _____________
iii. _____________ iv. _____________
4. Viruses live on __________ line of living and non-living. They do not own _______ apparatus. They enter the body of living cell and start ___________.
5. The living cell inside which the virus grows and multiplies is known as ________.
6. Study of viruses is called ___________.

Saturday 24 June 2017
CBSE Class 8 - Science - Coal and Petroleum (Q and A) (#cbseNotes)
Coal and Petroleum
(NCERT Exemplar Q & A)
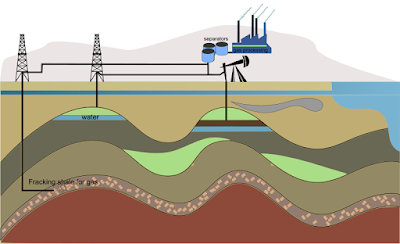 |
| Petroleum Refinery |
Question 1: You are provided with a mixture of petroleum and water. Can you suggest a method to separate the two?
Answer: Decantation
Question 2: What does CNG stand for and why is it considered to be a better fuel then petrol?
Answer: CNG stands for compressed natural gas. It is considered to be a better fuel because it is less polluting.
Question 3: Name the petroleum product used as fuel for stoves, lamps and jet aircrafts.
Answer: Kerosene is used as fuel for stove,lamps and jet aircrafts.
Question 4: Fill in the blanks in the following sentences.
1. Coal is one of the ________ used to cook food.
2. When heated in air, coal burns and produces mainly _________ gas.
3. Coal Tar is a black, thick - with an ________ smell.
4. Petroleum, ______ and _______ are fossil fuels.
5. Forest and coal are _________ natural resources.

Tuesday 13 June 2017
CBSE Class 8 - Science - CH4 Metals and Non-Metals (NCERT Exemplar Answers) (#cbseNotes)
Metals and Non-Metals
NCERT Exemplar Answers
Q1: Which of the following is not a metal?
(a) copper
(b) sulphur
(c) aluminium
(d) iron
Q2: The substance that will be flattened on beating with a hammer is
(a) crystal of iodine
(b) lump of sulphur
(c) piece of coal
(d) zinc granule

Friday 19 May 2017
CBSE Class 8/10 - Metals vs Non-Metals (Comparison) (#cbseNotes)
Metals vs Non-Metals
Physical Properties Comparison
| Property | Metals | Non Metals |
|---|---|---|
| Physical State | Most metals are solids at room temperature except mercury which is a liquid. Gallium and Cesium also changes into liquid at slightly above room temperature. | Non Metals exist in all states. Eleven are gases, bromine is liquid while others are solids. |
| Melting and Boiling Point | Generally have high melting and boiling points. (Exceptions are Gallium, Mercury, Sodium and Potassium.) | Generally they have low melting and boiling points. Exceptions are: Carbon, Boron and Silicon. |
| Density | They have high density except Sodium and potassium. | Low denisty (Diamond is an exception.) |
| Lusture | Metals shine and are lustrous. | Non-metalls appear dull. (Exceptions are Diamond, graphite and iodine.) |

Tuesday 16 May 2017
CBSE Class 8 - Science - CH 13 - Sound (Question Bank) (#cbseNotes)
SOUND
Question Bank
Very Short Answer Type Questions
Q1: Define vibration.
Q2: What is meant by oscillatory motion?
Q3: How is sound produced?
Q4: Can sound travel in vacuum?
Q5: Define amplitude.

Tuesday 9 May 2017
Sunday 19 March 2017
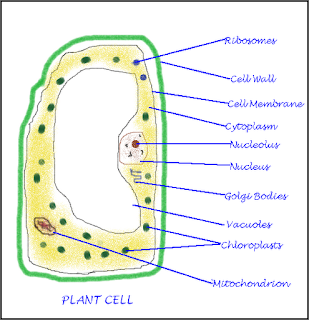
CBSE Class 8 - Biology - Cell Organelles, Location and Functions (#cbsenotes)
Cell Organelles, Location and Functions
| Organelles | Present In | Description | Function |
|---|---|---|---|
| Cell Wall | Plants only | Outer most covering, stiff shape, made of cellulose | Protects the cell from temperature variation, inside water pressure and moisture, procvides structural strength |
| Cell Membrane | Both plants and animals | Plants inside the cell wall. Animal: thin outermost selectively permeable |
Allows exchange of selected substance, supports the cell, separates the cell from its environment. |
| Nucleus | Both plants and animals | Oval shaped body | Control cell activities, transmits genetics characteristics |
| Cytoplasm | Both plants and animals | Thick jelly like fluid containing other cell organelles | Site of all life (metabolic) processes, protects cell organelles |
| Mitochondria | Both plants and animals | Rod shaped body | Site of respiration, releases energy form sugar, called power house of the cell |
| Golgi body | Both plants and animals | Parallel stacked vesicles | packaging and dispatch of proteins and lipids, helps in lysosomes formation |
| Endoplasmic reticulum | Both plants and animals | Networks of tubes and membrane | Transportation of materials. |
| Ribosomes | Both plants and animals | Small bodies, sometimes attached to endoplasmic reticulum | protein synthesis |
| Vacuole | Plant: single and large. Animal: very small or absent |
Sac-like structure in cytoplasm | Stores food and water (cell sap) |
| Plastids | Only Plants | Small colored bodies in cytoplasm | Impart color to leaf, flower and fruit |
| Lysosomes | Generally animals | Membrane bound sac in cytoplasm filled with digestive enzymes | Eats up(digests) damaged cells and unwanted cell products |
✎Note: See ☛ Class 8 Science - Cell (NCERT Q and A)
☛ Cell - MCQs
☛Cell Structure and Functions (Q and A)
☛ Cell - MCQs
☛Cell Structure and Functions (Q and A)
Friday 10 March 2017
Monday 27 February 2017
Wednesday 14 December 2016
CBSE Class 8 - Science - REACHING THE AGE OF ADOLESCENCE
REACHING THE AGE OF ADOLESCENCE
Q & A Based NCERT Class 8 Science Chapter
Q 1: List the changes occur during adolescent age.
Answer:
CHANGES OCCUR AT ADOLESCENT AGE:
1) INCREASE IN HEIGHT
2) DEVELOPMENT OF HAIR
3) SEXUAL FEELING
4) VOICE CHANGES
5) CHANGE IN BODY SHAPE
6) CHANGE IN BEHAVIOUR AND ATTITUDES
7) ACTIVITY OF SWEAT AND OIL GLANDS
Q 2: What is the function of endocrine system? What is it made up of?
Answer: There are a number of other slower processes that go on in the body, such as growth such processes are primarily controlled by the endocrine system.
The endocrine system consist of a number of ductless glands that secrete hormones.
Q3: What is Adam's apple?
Answer: Adam's apple: The protruding part of the throat is called Adam’s apple. It is the enlarged voice box or larynx which gets enlarged and visible from outside in boys at the onset of puberty. This makes the voice of boys hoarse.
Q 4: Name four body processes that hormones control?
Answer: Growth behavior, development and reproduction are controlled by hormones.

Friday 30 September 2016
Class 8, 10: Science - Uses of Some Common Metals and Non-Metals
Uses of Some Common Metals and Non-Metals
USES OF SOME COMMON METALS
| Metal | Main Uses |
|---|---|
| Iron | Bridges, Train tracks, Construction Bars, Steel, Engine Parts, Iron sheets etc. |
| Copper | Electrical Wires, Utensils, Coins etc. |
| Aluminium | Electric Wires, Utensils, Airplanes, Metallic Paints, Foil for packaging etc. |
| Silver | Jewellery, Electroplating, photography, Utensils, Coins, silvering of mirrors etc. |
| Gold | Jewellery and Decorations, Coins |
| Lead | Batteries, Alloys, Paints, Alloys like solder, Protective screen for X-Rays etc. |
| Mercury | Thermometers, Barometers, Dental amalgams etc. |

Friday 15 July 2016
CBSE Class 8 - Science - COMBUSTION AND FLAME (HOTS Questions and Answers)
COMBUSTION AND FLAME
 |
| Forest Fires - Why do forests catch fire in hot summers? |
HOTS Q & A (Class 8 Living Science Textbook Chapter Solutions)
Q1: Only gases burn with a flame. When you burn wood it initially burns with a flame. Later it only glows without a flame. What do you think the answer is?
Answer: Wood contains many chemicals like resin, lignin. When heated, these chemicals broke up and release combustible gases. That's why wood burns initially with a flame. When these gases are exhausted, wood burns without flame.
Q2: Would you consider coal as a good fuel? Give reasons?
Answer: Coal is not considered as a good fuel because of the following reasons:
1. It has low calorific value as compared to Petroleum and LPG.
2. On burning it releases many polluting gases like CO2 which is a green house gas and it causes global warming.
3. Coal contains traces of Sulphur and Nitrogen which on burning gives harmful gases like Nitrogen Oxide and Sulphur Oxide. These gases when mixed with rain water cause acid rain.
4. Unburnt traces of carbon also causes respiratory problems.
Q3: When a fuel is burnt, carbon dioxide (or carbon monoxide) and water vapour are given out. Can you name one fuel which burns without giving off water vapour? Why does it not give out water?
Answer: Charcoal is pure form of carbon. When it burns with oxygen, it forms carbon dioxide and no waters vapours are given off.
Q4: Why does a matchstick burn on rubbing it on the side of the match box?
Answer: Matchstick uses chemicals like phosphorus which has low ignition temperature (35°C). When the stick is rubbed, due to friction heat is generated which is sufficient to raise temperature to above 35°C and ignites the stick.

Subscribe to:
Posts (Atom)
