Alloys - Compositions, Properties and Uses
① Steel
Components: Iron, Carbon
Properties: Hard and Strong
Uses:
- Construction of buildings and bridges,
- manufacture of ships
- manufacture of spare parts for automobiles
Blog provides NCERT solutions, CBSE, NTSE, Olympiad study material, model test papers, important Questions and Answers asked in CBSE examinations. References to Educational Sites and resources.







 |
| Let's move the rock! |


 |
| Resin Coding System (Image Credits: Openclipart) |





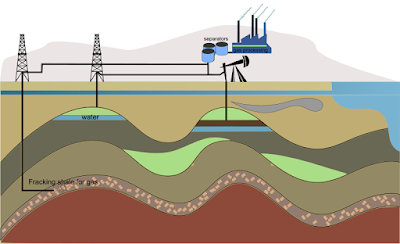 |
| Petroleum Refinery |


| Property | Metals | Non Metals |
|---|---|---|
| Physical State | Most metals are solids at room temperature except mercury which is a liquid. Gallium and Cesium also changes into liquid at slightly above room temperature. | Non Metals exist in all states. Eleven are gases, bromine is liquid while others are solids. |
| Melting and Boiling Point | Generally have high melting and boiling points. (Exceptions are Gallium, Mercury, Sodium and Potassium.) | Generally they have low melting and boiling points. Exceptions are: Carbon, Boron and Silicon. |
| Density | They have high density except Sodium and potassium. | Low denisty (Diamond is an exception.) |
| Lusture | Metals shine and are lustrous. | Non-metalls appear dull. (Exceptions are Diamond, graphite and iodine.) |


Please turn off the ad blocker. This is only way that we can earn some penny. Please support us by trun off the ad blocker.
Thank you!!